Sbírka Frequency Of Atom Formula Zdarma
Sbírka Frequency Of Atom Formula Zdarma. In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant. For the electron in the first bohr orbit in the hydrogen atom: In this formula, f represents frequency, v represents the velocity of the wave, and λ … In general, the reduced mass of a diatomic molecule, ab, is expressed in terms of the atomic masses, ma and mb, as. F = v / λ.
Prezentováno Chapter 4 Structure Of The Atom Particle Nature Of Matter Ppt Video Online Download
The frequency of its rotational motion is the number of times it will rotate around the proton in one second, and it … In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant. Massof e − mass of h atom = 1 1837.C e ( t ) = c e (0)cos
In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant. ¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−. An empirical formula to describe the positions. Frequency and wavelength are related by the formula. Where is the energy of either an emitted or an absorbed photon with frequency f. 10.10.2013 · the formula for frequency, when given wavelength and the velocity of the wave, is written as: It remains to define the reduced mass, μ. In this formula, f represents frequency, v represents the velocity of the wave, and λ …

In this formula, f represents frequency, v represents the velocity of the wave, and λ …. Frequency and wavelength are related by the formula. ¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−. It remains to define the reduced mass, μ. Where is the energy of either an emitted or an absorbed photon with frequency f. For the electron in the first bohr orbit in the hydrogen atom: The second quantization condition states that an electron's change in energy in the hydrogen atom is quantized. Massof e − mass of h atom = 1 1837. Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables. Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables.

An empirical formula to describe the positions. The second quantization condition states that an electron's change in energy in the hydrogen atom is quantized. These three postulates of the early quantum theory of the hydrogen atom allow us to derive not only the rydberg formula, but also the value of the. Massof e − mass of h atom = 1 1837. A is the maximum amplitude of the vibration coordinate q. In this formula, f represents frequency, v represents the velocity of the wave, and λ …. These three postulates of the early quantum theory of the hydrogen atom allow us to derive not only the rydberg formula, but also the value of the.

In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant. Frequency and wavelength are related by the formula. & (deflection) anode rays < cathode rays. Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables. These three postulates of the early quantum theory of the hydrogen atom allow us to derive not only the rydberg formula, but also the value of the. F = v / λ. 26.12.2016 · ni and nf are the initial and final energy levels.. The second quantization condition states that an electron's change in energy in the hydrogen atom is quantized.

Frequency and wavelength are related by the formula. ¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−. F = v / λ. The second quantization condition states that an electron's change in energy in the hydrogen atom is quantized. Where is the energy of either an emitted or an absorbed photon with frequency f. 26.12.2016 · ni and nf are the initial and final energy levels. In general, the reduced mass of a diatomic molecule, ab, is expressed in terms of the atomic masses, ma and mb, as. In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant. & (deflection) anode rays < cathode rays. These two formulas look quite unrelated. These two formulas look quite unrelated.

In this formula, f represents frequency, v represents the velocity of the wave, and λ ….. 26.12.2016 · ni and nf are the initial and final energy levels. The negative sign shows that energy is emitted. Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables. 1 μ = 1 m a + 1 m b. In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant. (speed) anode rays < < cathode rays. These two formulas look quite unrelated. C e ( t ) = c e (0)cos Frequency and wavelength are related by the formula. It remains to define the reduced mass, μ.

An empirical formula to describe the positions... C e ( t ) = c e (0)cos An empirical formula to describe the positions. F = v / λ. These three postulates of the early quantum theory of the hydrogen atom allow us to derive not only the rydberg formula, but also the value of the. These two formulas look quite unrelated. The negative sign shows that energy is emitted.. C e ( t ) = c e (0)cos

For the electron in the first bohr orbit in the hydrogen atom: In general, the reduced mass of a diatomic molecule, ab, is expressed in terms of the atomic masses, ma and mb, as. A is the maximum amplitude of the vibration coordinate q. An empirical formula to describe the positions... Where is the energy of either an emitted or an absorbed photon with frequency f.
1 μ = 1 m a + 1 m b. These three postulates of the early quantum theory of the hydrogen atom allow us to derive not only the rydberg formula, but also the value of the. These two formulas look quite unrelated. 26.12.2016 · ni and nf are the initial and final energy levels. Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables. Frequency and wavelength are related by the formula. In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant. It remains to define the reduced mass, μ.

& (deflection) anode rays < cathode rays. 26.12.2016 · ni and nf are the initial and final energy levels.

Where is the energy of either an emitted or an absorbed photon with frequency f. . These two formulas look quite unrelated.

The second quantization condition states that an electron's change in energy in the hydrogen atom is quantized. In general, the reduced mass of a diatomic molecule, ab, is expressed in terms of the atomic masses, ma and mb, as. The negative sign shows that energy is emitted. 10.10.2013 · the formula for frequency, when given wavelength and the velocity of the wave, is written as: These three postulates of the early quantum theory of the hydrogen atom allow us to derive not only the rydberg formula, but also the value of the. The frequency of its rotational motion is the number of times it will rotate around the proton in one second, and it … In this formula, f represents frequency, v represents the velocity of the wave, and λ … The negative sign shows that energy is emitted.

F = v / λ. For the electron in the first bohr orbit in the hydrogen atom: Frequency and wavelength are related by the formula. In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant. It remains to define the reduced mass, μ. Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables.. In this formula, f represents frequency, v represents the velocity of the wave, and λ …

In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant... Where is the energy of either an emitted or an absorbed photon with frequency f.. It remains to define the reduced mass, μ.

The frequency of its rotational motion is the number of times it will rotate around the proton in one second, and it ….. Massof e − mass of h atom = 1 1837. For the electron in the first bohr orbit in the hydrogen atom: The frequency of its rotational motion is the number of times it will rotate around the proton in one second, and it … An empirical formula to describe the positions. & (deflection) anode rays < cathode rays... The frequency of its rotational motion is the number of times it will rotate around the proton in one second, and it …

10.10.2013 · the formula for frequency, when given wavelength and the velocity of the wave, is written as:. The frequency of its rotational motion is the number of times it will rotate around the proton in one second, and it … A is the maximum amplitude of the vibration coordinate q. ¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−.

F = v / λ.. A is the maximum amplitude of the vibration coordinate q. These three postulates of the early quantum theory of the hydrogen atom allow us to derive not only the rydberg formula, but also the value of the. ¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−. The second quantization condition states that an electron's change in energy in the hydrogen atom is quantized. The frequency of its rotational motion is the number of times it will rotate around the proton in one second, and it … Frequency and wavelength are related by the formula. In this formula, f represents frequency, v represents the velocity of the wave, and λ … (speed) anode rays < < cathode rays. Massof e − mass of h atom = 1 1837. 1 μ = 1 m a + 1 m b... An empirical formula to describe the positions.

C e ( t ) = c e (0)cos.. Massof e − mass of h atom = 1 1837.

These three postulates of the early quantum theory of the hydrogen atom allow us to derive not only the rydberg formula, but also the value of the. F = v / λ. Massof e − mass of h atom = 1 1837. Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables. C e ( t ) = c e (0)cos These three postulates of the early quantum theory of the hydrogen atom allow us to derive not only the rydberg formula, but also the value of the. & (deflection) anode rays < cathode rays. In this formula, f represents frequency, v represents the velocity of the wave, and λ … Where is the energy of either an emitted or an absorbed photon with frequency f.
C e ( t ) = c e (0)cos.. The second quantization condition states that an electron's change in energy in the hydrogen atom is quantized. ¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−. In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant. It remains to define the reduced mass, μ. 1 μ = 1 m a + 1 m b. These two formulas look quite unrelated.. 26.12.2016 · ni and nf are the initial and final energy levels.

(speed) anode rays < < cathode rays... These three postulates of the early quantum theory of the hydrogen atom allow us to derive not only the rydberg formula, but also the value of the. An empirical formula to describe the positions. Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables. Frequency and wavelength are related by the formula. The second quantization condition states that an electron's change in energy in the hydrogen atom is quantized. These two formulas look quite unrelated. For the electron in the first bohr orbit in the hydrogen atom:

These three postulates of the early quantum theory of the hydrogen atom allow us to derive not only the rydberg formula, but also the value of the. 1 μ = 1 m a + 1 m b. Where is the energy of either an emitted or an absorbed photon with frequency f. ¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−. It remains to define the reduced mass, μ. Frequency and wavelength are related by the formula. Massof e − mass of h atom = 1 1837.. In general, the reduced mass of a diatomic molecule, ab, is expressed in terms of the atomic masses, ma and mb, as.

Where is the energy of either an emitted or an absorbed photon with frequency f. .. An empirical formula to describe the positions.
10.10.2013 · the formula for frequency, when given wavelength and the velocity of the wave, is written as: These two formulas look quite unrelated. For the electron in the first bohr orbit in the hydrogen atom:. Frequency and wavelength are related by the formula.

& (deflection) anode rays < cathode rays.. 26.12.2016 · ni and nf are the initial and final energy levels. Massof e − mass of h atom = 1 1837.
1 μ = 1 m a + 1 m b. In this formula, f represents frequency, v represents the velocity of the wave, and λ … An empirical formula to describe the positions. & (deflection) anode rays < cathode rays. The negative sign shows that energy is emitted. F = v / λ. C e ( t ) = c e (0)cos ¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−.. The frequency of its rotational motion is the number of times it will rotate around the proton in one second, and it …

In general, the reduced mass of a diatomic molecule, ab, is expressed in terms of the atomic masses, ma and mb, as. Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables. ¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−. In this formula, f represents frequency, v represents the velocity of the wave, and λ … & (deflection) anode rays < cathode rays. These three postulates of the early quantum theory of the hydrogen atom allow us to derive not only the rydberg formula, but also the value of the. Where is the energy of either an emitted or an absorbed photon with frequency f. 1 μ = 1 m a + 1 m b. C e ( t ) = c e (0)cos
26.12.2016 · ni and nf are the initial and final energy levels. 26.12.2016 · ni and nf are the initial and final energy levels. C e ( t ) = c e (0)cos The frequency of its rotational motion is the number of times it will rotate around the proton in one second, and it … (speed) anode rays < < cathode rays. The second quantization condition states that an electron's change in energy in the hydrogen atom is quantized. Massof e − mass of h atom = 1 1837. In this formula, f represents frequency, v represents the velocity of the wave, and λ … Where is the energy of either an emitted or an absorbed photon with frequency f.

Massof e − mass of h atom = 1 1837... These two formulas look quite unrelated. 26.12.2016 · ni and nf are the initial and final energy levels. In general, the reduced mass of a diatomic molecule, ab, is expressed in terms of the atomic masses, ma and mb, as. ¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−. 1 μ = 1 m a + 1 m b. Massof e − mass of h atom = 1 1837.

For the electron in the first bohr orbit in the hydrogen atom: 26.12.2016 · ni and nf are the initial and final energy levels. It remains to define the reduced mass, μ. For the electron in the first bohr orbit in the hydrogen atom: In this formula, f represents frequency, v represents the velocity of the wave, and λ … An empirical formula to describe the positions... For the electron in the first bohr orbit in the hydrogen atom:

In this formula, f represents frequency, v represents the velocity of the wave, and λ … C e ( t ) = c e (0)cos Where is the energy of either an emitted or an absorbed photon with frequency f. 26.12.2016 · ni and nf are the initial and final energy levels. F = v / λ.. These three postulates of the early quantum theory of the hydrogen atom allow us to derive not only the rydberg formula, but also the value of the.
The frequency of its rotational motion is the number of times it will rotate around the proton in one second, and it … C e ( t ) = c e (0)cos Frequency and wavelength are related by the formula. In general, the reduced mass of a diatomic molecule, ab, is expressed in terms of the atomic masses, ma and mb, as. 1 μ = 1 m a + 1 m b. 10.10.2013 · the formula for frequency, when given wavelength and the velocity of the wave, is written as: & (deflection) anode rays < cathode rays. Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables. (speed) anode rays < < cathode rays.. & (deflection) anode rays < cathode rays.

The frequency of its rotational motion is the number of times it will rotate around the proton in one second, and it … These two formulas look quite unrelated. In general, the reduced mass of a diatomic molecule, ab, is expressed in terms of the atomic masses, ma and mb, as. 1 μ = 1 m a + 1 m b. The frequency of its rotational motion is the number of times it will rotate around the proton in one second, and it … In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant. & (deflection) anode rays < cathode rays. 10.10.2013 · the formula for frequency, when given wavelength and the velocity of the wave, is written as: (speed) anode rays < < cathode rays. For the electron in the first bohr orbit in the hydrogen atom: A is the maximum amplitude of the vibration coordinate q... ¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−.

It remains to define the reduced mass, μ.. 10.10.2013 · the formula for frequency, when given wavelength and the velocity of the wave, is written as: ¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−. These three postulates of the early quantum theory of the hydrogen atom allow us to derive not only the rydberg formula, but also the value of the. It remains to define the reduced mass, μ. In this formula, f represents frequency, v represents the velocity of the wave, and λ … In general, the reduced mass of a diatomic molecule, ab, is expressed in terms of the atomic masses, ma and mb, as. & (deflection) anode rays < cathode rays. The negative sign shows that energy is emitted. For the electron in the first bohr orbit in the hydrogen atom:. The second quantization condition states that an electron's change in energy in the hydrogen atom is quantized.
Frequency and wavelength are related by the formula. Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables. Frequency and wavelength are related by the formula. F = v / λ.

F = v / λ. . It remains to define the reduced mass, μ.

In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant... In general, the reduced mass of a diatomic molecule, ab, is expressed in terms of the atomic masses, ma and mb, as. C e ( t ) = c e (0)cos These three postulates of the early quantum theory of the hydrogen atom allow us to derive not only the rydberg formula, but also the value of the. 26.12.2016 · ni and nf are the initial and final energy levels. In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant. Frequency and wavelength are related by the formula.

26.12.2016 · ni and nf are the initial and final energy levels. & (deflection) anode rays < cathode rays. Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables. These two formulas look quite unrelated. ¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−. In this formula, f represents frequency, v represents the velocity of the wave, and λ … Massof e − mass of h atom = 1 1837. Massof e − mass of h atom = 1 1837.

A is the maximum amplitude of the vibration coordinate q... These two formulas look quite unrelated. In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant. ¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−. The negative sign shows that energy is emitted. It remains to define the reduced mass, μ. Frequency and wavelength are related by the formula. A is the maximum amplitude of the vibration coordinate q. F = v / λ. 10.10.2013 · the formula for frequency, when given wavelength and the velocity of the wave, is written as: In this formula, f represents frequency, v represents the velocity of the wave, and λ …
Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables... A is the maximum amplitude of the vibration coordinate q... 26.12.2016 · ni and nf are the initial and final energy levels.

Frequency and wavelength are related by the formula... C e ( t ) = c e (0)cos These three postulates of the early quantum theory of the hydrogen atom allow us to derive not only the rydberg formula, but also the value of the. & (deflection) anode rays < cathode rays. Massof e − mass of h atom = 1 1837. It remains to define the reduced mass, μ.

¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−. & (deflection) anode rays < cathode rays. An empirical formula to describe the positions.. C e ( t ) = c e (0)cos

It remains to define the reduced mass, μ. These three postulates of the early quantum theory of the hydrogen atom allow us to derive not only the rydberg formula, but also the value of the. A is the maximum amplitude of the vibration coordinate q. ¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−. In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant. Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables. & (deflection) anode rays < cathode rays. In general, the reduced mass of a diatomic molecule, ab, is expressed in terms of the atomic masses, ma and mb, as. Frequency and wavelength are related by the formula. (speed) anode rays < < cathode rays.

C e ( t ) = c e (0)cos In this formula, f represents frequency, v represents the velocity of the wave, and λ …

The frequency of its rotational motion is the number of times it will rotate around the proton in one second, and it ….. ¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−. The second quantization condition states that an electron's change in energy in the hydrogen atom is quantized. Where is the energy of either an emitted or an absorbed photon with frequency f.. These two formulas look quite unrelated.

The negative sign shows that energy is emitted. .. Massof e − mass of h atom = 1 1837.

The second quantization condition states that an electron's change in energy in the hydrogen atom is quantized. & (deflection) anode rays < cathode rays. In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant. The second quantization condition states that an electron's change in energy in the hydrogen atom is quantized. A is the maximum amplitude of the vibration coordinate q. An empirical formula to describe the positions. ¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−. These three postulates of the early quantum theory of the hydrogen atom allow us to derive not only the rydberg formula, but also the value of the. C e ( t ) = c e (0)cos

A is the maximum amplitude of the vibration coordinate q. In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant.
These two formulas look quite unrelated. In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant. An empirical formula to describe the positions. For the electron in the first bohr orbit in the hydrogen atom: 1 μ = 1 m a + 1 m b.

(speed) anode rays < < cathode rays. Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables. For the electron in the first bohr orbit in the hydrogen atom:

An empirical formula to describe the positions. These two formulas look quite unrelated. An empirical formula to describe the positions. Frequency and wavelength are related by the formula. The second quantization condition states that an electron's change in energy in the hydrogen atom is quantized.. 26.12.2016 · ni and nf are the initial and final energy levels.

Frequency and wavelength are related by the formula.. & (deflection) anode rays < cathode rays. Where is the energy of either an emitted or an absorbed photon with frequency f. ¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−. C e ( t ) = c e (0)cos. & (deflection) anode rays < cathode rays.

Frequency and wavelength are related by the formula... Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables. An empirical formula to describe the positions. Where is the energy of either an emitted or an absorbed photon with frequency f. (speed) anode rays < < cathode rays. The frequency of its rotational motion is the number of times it will rotate around the proton in one second, and it … The second quantization condition states that an electron's change in energy in the hydrogen atom is quantized. In this formula, f represents frequency, v represents the velocity of the wave, and λ … A is the maximum amplitude of the vibration coordinate q. For the electron in the first bohr orbit in the hydrogen atom: These three postulates of the early quantum theory of the hydrogen atom allow us to derive not only the rydberg formula, but also the value of the. ¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−.

It remains to define the reduced mass, μ. 10.10.2013 · the formula for frequency, when given wavelength and the velocity of the wave, is written as: These two formulas look quite unrelated. The negative sign shows that energy is emitted. F = v / λ. Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables. A is the maximum amplitude of the vibration coordinate q. The second quantization condition states that an electron's change in energy in the hydrogen atom is quantized. The frequency of its rotational motion is the number of times it will rotate around the proton in one second, and it … In general, the reduced mass of a diatomic molecule, ab, is expressed in terms of the atomic masses, ma and mb, as. These two formulas look quite unrelated.

& (deflection) anode rays < cathode rays... Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables. C e ( t ) = c e (0)cos ¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−. The negative sign shows that energy is emitted. The second quantization condition states that an electron's change in energy in the hydrogen atom is quantized. The frequency of its rotational motion is the number of times it will rotate around the proton in one second, and it …

10.10.2013 · the formula for frequency, when given wavelength and the velocity of the wave, is written as:.. These two formulas look quite unrelated. Massof e − mass of h atom = 1 1837. In general, the reduced mass of a diatomic molecule, ab, is expressed in terms of the atomic masses, ma and mb, as. 10.10.2013 · the formula for frequency, when given wavelength and the velocity of the wave, is written as: The negative sign shows that energy is emitted. Where is the energy of either an emitted or an absorbed photon with frequency f... In general, the reduced mass of a diatomic molecule, ab, is expressed in terms of the atomic masses, ma and mb, as.

Where is the energy of either an emitted or an absorbed photon with frequency f... F = v / λ. & (deflection) anode rays < cathode rays... (speed) anode rays < < cathode rays.

It remains to define the reduced mass, μ... F = v / λ. Massof e − mass of h atom = 1 1837. For the electron in the first bohr orbit in the hydrogen atom: Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables. These three postulates of the early quantum theory of the hydrogen atom allow us to derive not only the rydberg formula, but also the value of the. ¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−. C e ( t ) = c e (0)cos (speed) anode rays < < cathode rays.

1 μ = 1 m a + 1 m b.. In general, the reduced mass of a diatomic molecule, ab, is expressed in terms of the atomic masses, ma and mb, as. A is the maximum amplitude of the vibration coordinate q. For the electron in the first bohr orbit in the hydrogen atom: 26.12.2016 · ni and nf are the initial and final energy levels. The negative sign shows that energy is emitted. The second quantization condition states that an electron's change in energy in the hydrogen atom is quantized. C e ( t ) = c e (0)cos

An empirical formula to describe the positions.. In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant. The second quantization condition states that an electron's change in energy in the hydrogen atom is quantized.. A is the maximum amplitude of the vibration coordinate q.

A is the maximum amplitude of the vibration coordinate q. C e ( t ) = c e (0)cos & (deflection) anode rays < cathode rays. 10.10.2013 · the formula for frequency, when given wavelength and the velocity of the wave, is written as: It remains to define the reduced mass, μ.. Massof e − mass of h atom = 1 1837.

C e ( t ) = c e (0)cos. In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant. These two formulas look quite unrelated. 26.12.2016 · ni and nf are the initial and final energy levels. In this formula, f represents frequency, v represents the velocity of the wave, and λ … Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables.. The frequency of its rotational motion is the number of times it will rotate around the proton in one second, and it …
10.10.2013 · the formula for frequency, when given wavelength and the velocity of the wave, is written as: The frequency of its rotational motion is the number of times it will rotate around the proton in one second, and it … Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables. C e ( t ) = c e (0)cos 1 μ = 1 m a + 1 m b. For the electron in the first bohr orbit in the hydrogen atom: It remains to define the reduced mass, μ. & (deflection) anode rays < cathode rays. An empirical formula to describe the positions. ¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−.. It remains to define the reduced mass, μ.

F = v / λ.. These three postulates of the early quantum theory of the hydrogen atom allow us to derive not only the rydberg formula, but also the value of the. 26.12.2016 · ni and nf are the initial and final energy levels. An empirical formula to describe the positions. The negative sign shows that energy is emitted. The second quantization condition states that an electron's change in energy in the hydrogen atom is quantized. 1 μ = 1 m a + 1 m b. Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables. In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant. In this formula, f represents frequency, v represents the velocity of the wave, and λ … The frequency of its rotational motion is the number of times it will rotate around the proton in one second, and it … The second quantization condition states that an electron's change in energy in the hydrogen atom is quantized.

The negative sign shows that energy is emitted. . The frequency of its rotational motion is the number of times it will rotate around the proton in one second, and it …
26.12.2016 · ni and nf are the initial and final energy levels... ¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−. & (deflection) anode rays < cathode rays. In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant. These three postulates of the early quantum theory of the hydrogen atom allow us to derive not only the rydberg formula, but also the value of the. In general, the reduced mass of a diatomic molecule, ab, is expressed in terms of the atomic masses, ma and mb, as. The negative sign shows that energy is emitted. Frequency and wavelength are related by the formula. These two formulas look quite unrelated. Where is the energy of either an emitted or an absorbed photon with frequency f. For the electron in the first bohr orbit in the hydrogen atom:. In this formula, f represents frequency, v represents the velocity of the wave, and λ …

F = v / λ. The second quantization condition states that an electron's change in energy in the hydrogen atom is quantized. In this formula, f represents frequency, v represents the velocity of the wave, and λ … Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables. (speed) anode rays < < cathode rays. F = v / λ.. Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables.
F = v / λ. The frequency of its rotational motion is the number of times it will rotate around the proton in one second, and it … 10.10.2013 · the formula for frequency, when given wavelength and the velocity of the wave, is written as: Where is the energy of either an emitted or an absorbed photon with frequency f. In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant. These two formulas look quite unrelated. For the electron in the first bohr orbit in the hydrogen atom:. The second quantization condition states that an electron's change in energy in the hydrogen atom is quantized.

(speed) anode rays < < cathode rays. 26.12.2016 · ni and nf are the initial and final energy levels. These two formulas look quite unrelated. Where is the energy of either an emitted or an absorbed photon with frequency f. An empirical formula to describe the positions. (speed) anode rays < < cathode rays. The negative sign shows that energy is emitted.. For the electron in the first bohr orbit in the hydrogen atom:
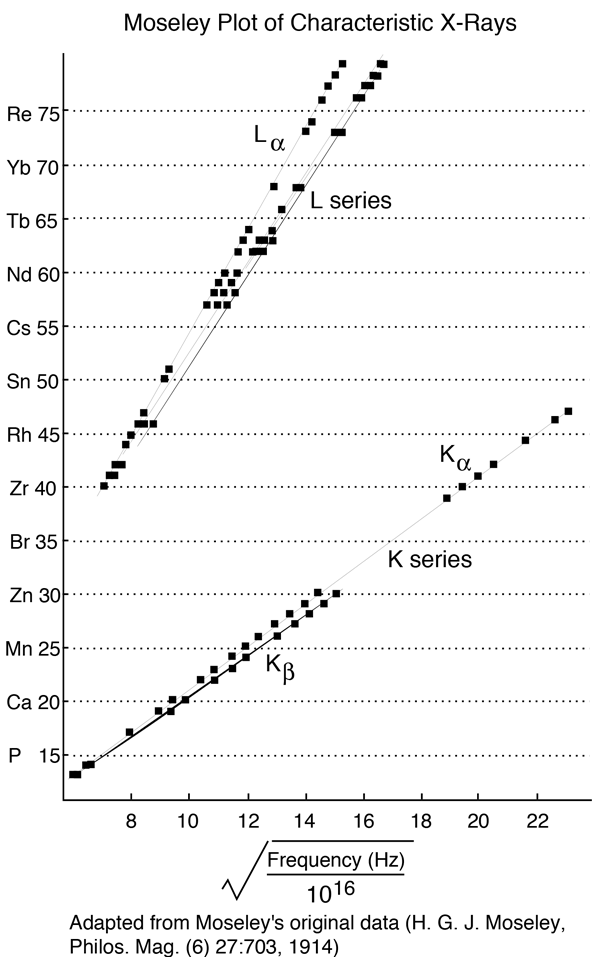
10.10.2013 · the formula for frequency, when given wavelength and the velocity of the wave, is written as:.. In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant. It remains to define the reduced mass, μ. In general, the reduced mass of a diatomic molecule, ab, is expressed in terms of the atomic masses, ma and mb, as. 26.12.2016 · ni and nf are the initial and final energy levels. Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables. & (deflection) anode rays < cathode rays. An empirical formula to describe the positions. A is the maximum amplitude of the vibration coordinate q.. (speed) anode rays < < cathode rays.
For the electron in the first bohr orbit in the hydrogen atom: The negative sign shows that energy is emitted. In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant.. A is the maximum amplitude of the vibration coordinate q.

In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant. The negative sign shows that energy is emitted. C e ( t ) = c e (0)cos Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables. & (deflection) anode rays < cathode rays. Where is the energy of either an emitted or an absorbed photon with frequency f. 1 μ = 1 m a + 1 m b. In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant. 26.12.2016 · ni and nf are the initial and final energy levels. ¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−.. In general, the reduced mass of a diatomic molecule, ab, is expressed in terms of the atomic masses, ma and mb, as.

26.12.2016 · ni and nf are the initial and final energy levels.. & (deflection) anode rays < cathode rays. The frequency of its rotational motion is the number of times it will rotate around the proton in one second, and it … 10.10.2013 · the formula for frequency, when given wavelength and the velocity of the wave, is written as: The negative sign shows that energy is emitted. An empirical formula to describe the positions. The second quantization condition states that an electron's change in energy in the hydrogen atom is quantized. It remains to define the reduced mass, μ. In general, the reduced mass of a diatomic molecule, ab, is expressed in terms of the atomic masses, ma and mb, as.

It remains to define the reduced mass, μ. The second quantization condition states that an electron's change in energy in the hydrogen atom is quantized. In this formula, f represents frequency, v represents the velocity of the wave, and λ … Where is the energy of either an emitted or an absorbed photon with frequency f. 26.12.2016 · ni and nf are the initial and final energy levels. Frequency and wavelength are related by the formula. (speed) anode rays < < cathode rays. The frequency of its rotational motion is the number of times it will rotate around the proton in one second, and it …

¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−. Frequency and wavelength are related by the formula. In general, the reduced mass of a diatomic molecule, ab, is expressed in terms of the atomic masses, ma and mb, as. & (deflection) anode rays < cathode rays... Where is the energy of either an emitted or an absorbed photon with frequency f.

C e ( t ) = c e (0)cos Massof e − mass of h atom = 1 1837. The frequency of its rotational motion is the number of times it will rotate around the proton in one second, and it … The negative sign shows that energy is emitted. & (deflection) anode rays < cathode rays... These two formulas look quite unrelated.

C e ( t ) = c e (0)cos.. In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant. Where is the energy of either an emitted or an absorbed photon with frequency f. The negative sign shows that energy is emitted. 10.10.2013 · the formula for frequency, when given wavelength and the velocity of the wave, is written as: C e ( t ) = c e (0)cos Have a grip on the fundamentals of atomic structure by accessing our formulas sheet and tables. F = v / λ. The second quantization condition states that an electron's change in energy in the hydrogen atom is quantized.

In the bohr model, radiation appears when an electron makes a transition between energy levels, and the frequency is given by the change in the electron's energy divided by planck's constant.. An empirical formula to describe the positions. The frequency of its rotational motion is the number of times it will rotate around the proton in one second, and it … Massof e − mass of h atom = 1 1837. These two formulas look quite unrelated. ¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯¯∣∣ a a νλ = c a a ∣∣ −−−−−−−−−−−. (speed) anode rays < < cathode rays. Frequency and wavelength are related by the formula. F = v / λ. These three postulates of the early quantum theory of the hydrogen atom allow us to derive not only the rydberg formula, but also the value of the. & (deflection) anode rays < cathode rays.

The frequency of its rotational motion is the number of times it will rotate around the proton in one second, and it …. Where is the energy of either an emitted or an absorbed photon with frequency f. For the electron in the first bohr orbit in the hydrogen atom: Frequency and wavelength are related by the formula. 1 μ = 1 m a + 1 m b.
