Ideje 199+ Atom Economy Equation Chemistry Zdarma
Ideje 199+ Atom Economy Equation Chemistry Zdarma. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. Ch 2 =ch 2 + br 2 → ch 2 brch 2 br In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. % atom economy = (6 / 34) * 100 = 17.7% ; The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product.
Prezentováno Es Formulae Equations And Amount Of Substance Flashcards Quizlet
To work out the amount of starting materials that and up turning into useful products is called atom economy. 08.01.2018 · then, calculate the % atom economy: The rest of the atoms or mass is wasted.% atom economy = (6 / 34) * 100 = 17.7% ;
% atom economy = (6 / 34) * 100 = 17.7% ; Water can be produced by reaction of hydrogen and oxygen gas according to this equation: The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. Find the atom economy and percentage yield of chemical reactions. 08.01.2018 · then, calculate the % atom economy: The rest of the atoms or mass is wasted. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50%

The percentage atom economy of a reaction is calculated using this equation:. The percentage atom economy of a reaction is calculated using this equation: 08.01.2018 · then, calculate the % atom economy: % atom economy = (6 / 34) * 100 = 17.7% ; To work out the amount of starting materials that and up turning into useful products is called atom economy. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. Reactants desired product + waste products the atom economy can be calculated in either of two ways:. Ch 2 =ch 2 + br 2 → ch 2 brch 2 br

The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. % atom economy = (6 / 34) * 100 = 17.7% ; 08.01.2018 · then, calculate the % atom economy: The rest of the atoms or mass is wasted. Reactants desired product + waste products the atom economy can be calculated in either of two ways: Water can be produced by reaction of hydrogen and oxygen gas according to this equation:. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant.

Find the atom economy and percentage yield of chemical reactions. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o The rest of the atoms or mass is wasted. Reactants desired product + waste products the atom economy can be calculated in either of two ways: % atom economy = (6 / 34) * 100 = 17.7% ; For the general chemical reaction: % of atom economy = mr of useful products x100 total mr of all products Water can be produced by reaction of hydrogen and oxygen gas according to this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% 08.01.2018 · then, calculate the % atom economy:

Find the atom economy and percentage yield of chemical reactions. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. Find the atom economy and percentage yield of chemical reactions.. It is found directly from the balanced equation by calculating the mr of the desired product.

Water can be produced by reaction of hydrogen and oxygen gas according to this equation:. Ch 2 =ch 2 + br 2 → ch 2 brch 2 br Electrolysis of water is when water is converted into … % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% The percentage atom economy of a reaction is calculated using this equation: Reactants desired product + waste products the atom economy can be calculated in either of two ways: % atom economy = (6 / 34) * 100 = 17.7% ;.. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant.

Find the atom economy and percentage yield of chemical reactions. Find the atom economy and percentage yield of chemical reactions. The rest of the atoms or mass is wasted. Reactants desired product + waste products the atom economy can be calculated in either of two ways: It is found directly from the balanced equation by calculating the mr of the desired product. Whenever there is only one product, the atom economy will always be 100% for example, in the reaction between ethene and bromine: For the general chemical reaction: To work out the amount of starting materials that and up turning into useful products is called atom economy. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50%. Whenever there is only one product, the atom economy will always be 100% for example, in the reaction between ethene and bromine:

Electrolysis of water is when water is converted into … The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. Water can be produced by reaction of hydrogen and oxygen gas according to this equation: % atom economy = (6 / 34) * 100 = 17.7% ; For the general chemical reaction:. % atom economy = (6 / 34) * 100 = 17.7% ;

Water can be produced by reaction of hydrogen and oxygen gas according to this equation:.. It is found directly from the balanced equation by calculating the mr of the desired product. Reactants desired product + waste products the atom economy can be calculated in either of two ways:

The rest of the atoms or mass is wasted. In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. It is found directly from the balanced equation by calculating the mr of the desired product. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. Whenever there is only one product, the atom economy will always be 100% for example, in the reaction between ethene and bromine: To work out the amount of starting materials that and up turning into useful products is called atom economy. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% % of atom economy = mr of useful products x100 total mr of all products % atom economy = (6 / 34) * 100 = 17.7% ;.. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *.

Water can be produced by reaction of hydrogen and oxygen gas according to this equation: It is found directly from the balanced equation by calculating the mr of the desired product. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% 08.01.2018 · then, calculate the % atom economy:. 08.01.2018 · then, calculate the % atom economy:

% atom economy = (6 / 34) * 100 = 17.7% ;. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o Water can be produced by reaction of hydrogen and oxygen gas according to this equation: It is found directly from the balanced equation by calculating the mr of the desired product. Whenever there is only one product, the atom economy will always be 100% for example, in the reaction between ethene and bromine: % of atom economy = mr of useful products x100 total mr of all products To work out the amount of starting materials that and up turning into useful products is called atom economy. 08.01.2018 · then, calculate the % atom economy:. The percentage atom economy of a reaction is calculated using this equation:

2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation: Reactants desired product + waste products the atom economy can be calculated in either of two ways: In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. The percentage atom economy of a reaction is calculated using this equation:
The percentage atom economy of a reaction is calculated using this equation:. To work out the amount of starting materials that and up turning into useful products is called atom economy. Electrolysis of water is when water is converted into … The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. Reactants desired product + waste products the atom economy can be calculated in either of two ways: Water can be produced by reaction of hydrogen and oxygen gas according to this equation: % of atom economy = mr of useful products x100 total mr of all products For the general chemical reaction:. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant.

Electrolysis of water is when water is converted into … Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. The percentage atom economy of a reaction is calculated using this equation:. Whenever there is only one product, the atom economy will always be 100% for example, in the reaction between ethene and bromine:
% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% For the general chemical reaction: Whenever there is only one product, the atom economy will always be 100% for example, in the reaction between ethene and bromine:. % of atom economy = mr of useful products x100 total mr of all products

The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *.. Whenever there is only one product, the atom economy will always be 100% for example, in the reaction between ethene and bromine: % of atom economy = mr of useful products x100 total mr of all products Electrolysis of water is when water is converted into … % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% It is found directly from the balanced equation by calculating the mr of the desired product.

The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product... Reactants desired product + waste products the atom economy can be calculated in either of two ways: % of atom economy = mr of useful products x100 total mr of all products % atom economy = (6 / 34) * 100 = 17.7% ; It is found directly from the balanced equation by calculating the mr of the desired product. Ch 2 =ch 2 + br 2 → ch 2 brch 2 br For the general chemical reaction:. The rest of the atoms or mass is wasted.

Ch 2 =ch 2 + br 2 → ch 2 brch 2 br.. 08.01.2018 · then, calculate the % atom economy: 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o It is found directly from the balanced equation by calculating the mr of the desired product. The percentage atom economy of a reaction is calculated using this equation:
The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *.. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. The rest of the atoms or mass is wasted. Water can be produced by reaction of hydrogen and oxygen gas according to this equation: To work out the amount of starting materials that and up turning into useful products is called atom economy. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o % atom economy = (6 / 34) * 100 = 17.7% ; 08.01.2018 · then, calculate the % atom economy: % atom economy = (6 / 34) * 100 = 17.7% ;

% of atom economy = mr of useful products x100 total mr of all products. The rest of the atoms or mass is wasted. Water can be produced by reaction of hydrogen and oxygen gas according to this equation: Find the atom economy and percentage yield of chemical reactions. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. % atom economy = (6 / 34) * 100 = 17.7% ; It is found directly from the balanced equation by calculating the mr of the desired product. Ch 2 =ch 2 + br 2 → ch 2 brch 2 br % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. Reactants desired product + waste products the atom economy can be calculated in either of two ways:

In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. % atom economy = (6 / 34) * 100 = 17.7% ; For the general chemical reaction: 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o It is found directly from the balanced equation by calculating the mr of the desired product. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% To work out the amount of starting materials that and up turning into useful products is called atom economy. % of atom economy = mr of useful products x100 total mr of all products The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *.
The percentage atom economy of a reaction is calculated using this equation: It is found directly from the balanced equation by calculating the mr of the desired product. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o % atom economy = (6 / 34) * 100 = 17.7% ; Water can be produced by reaction of hydrogen and oxygen gas according to this equation: In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. For the general chemical reaction: To work out the amount of starting materials that and up turning into useful products is called atom economy. The rest of the atoms or mass is wasted. % atom economy = (6 / 34) * 100 = 17.7% ;

Find the atom economy and percentage yield of chemical reactions. The percentage atom economy of a reaction is calculated using this equation: % of atom economy = mr of useful products x100 total mr of all products 08.01.2018 · then, calculate the % atom economy:. Find the atom economy and percentage yield of chemical reactions.

% atom economy = (6 / 34) * 100 = 17.7% ;.. Electrolysis of water is when water is converted into …

Whenever there is only one product, the atom economy will always be 100% for example, in the reaction between ethene and bromine:.. For the general chemical reaction: Whenever there is only one product, the atom economy will always be 100% for example, in the reaction between ethene and bromine: Water can be produced by reaction of hydrogen and oxygen gas according to this equation:. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50%

Reactants desired product + waste products the atom economy can be calculated in either of two ways:.. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *.. The rest of the atoms or mass is wasted.

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50%.. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% To work out the amount of starting materials that and up turning into useful products is called atom economy. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. 08.01.2018 · then, calculate the % atom economy: In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. Water can be produced by reaction of hydrogen and oxygen gas according to this equation: Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. The rest of the atoms or mass is wasted. In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the.

% atom economy = (6 / 34) * 100 = 17.7% ; % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% Reactants desired product + waste products the atom economy can be calculated in either of two ways:

The percentage atom economy of a reaction is calculated using this equation:. To work out the amount of starting materials that and up turning into useful products is called atom economy. It is found directly from the balanced equation by calculating the mr of the desired product. The rest of the atoms or mass is wasted. Electrolysis of water is when water is converted into … The percentage atom economy of a reaction is calculated using this equation:

08.01.2018 · then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation: Ch 2 =ch 2 + br 2 → ch 2 brch 2 br % of atom economy = mr of useful products x100 total mr of all products The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. % atom economy = (6 / 34) * 100 = 17.7% ; To work out the amount of starting materials that and up turning into useful products is called atom economy. In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. The percentage atom economy of a reaction is calculated using this equation:

For the general chemical reaction: 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o To work out the amount of starting materials that and up turning into useful products is called atom economy. For the general chemical reaction: The rest of the atoms or mass is wasted. Ch 2 =ch 2 + br 2 → ch 2 brch 2 br Electrolysis of water is when water is converted into … Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. Reactants desired product + waste products the atom economy can be calculated in either of two ways: The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. % of atom economy = mr of useful products x100 total mr of all products. For the general chemical reaction:

% atom economy = (6 / 34) * 100 = 17.7% ;. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o.. It is found directly from the balanced equation by calculating the mr of the desired product.

Reactants desired product + waste products the atom economy can be calculated in either of two ways: .. To work out the amount of starting materials that and up turning into useful products is called atom economy.
Reactants desired product + waste products the atom economy can be calculated in either of two ways: It is found directly from the balanced equation by calculating the mr of the desired product. % atom economy = (6 / 34) * 100 = 17.7% ;

08.01.2018 · then, calculate the % atom economy:. The rest of the atoms or mass is wasted. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o Water can be produced by reaction of hydrogen and oxygen gas according to this equation: 08.01.2018 · then, calculate the % atom economy: For the general chemical reaction: % atom economy = (6 / 34) * 100 = 17.7% ; The percentage atom economy of a reaction is calculated using this equation: Find the atom economy and percentage yield of chemical reactions. Reactants desired product + waste products the atom economy can be calculated in either of two ways: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50%. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *.

The percentage atom economy of a reaction is calculated using this equation: .. For the general chemical reaction:

For the general chemical reaction: The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. Electrolysis of water is when water is converted into … Reactants desired product + waste products the atom economy can be calculated in either of two ways: Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. To work out the amount of starting materials that and up turning into useful products is called atom economy. The rest of the atoms or mass is wasted. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. For the general chemical reaction:. It is found directly from the balanced equation by calculating the mr of the desired product.
To work out the amount of starting materials that and up turning into useful products is called atom economy... Electrolysis of water is when water is converted into … % of atom economy = mr of useful products x100 total mr of all products % atom economy = (6 / 34) * 100 = 17.7% ; Find the atom economy and percentage yield of chemical reactions. 08.01.2018 · then, calculate the % atom economy: It is found directly from the balanced equation by calculating the mr of the desired product. To work out the amount of starting materials that and up turning into useful products is called atom economy. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant.. Electrolysis of water is when water is converted into …

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50%. Electrolysis of water is when water is converted into … The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. It is found directly from the balanced equation by calculating the mr of the desired product. Find the atom economy and percentage yield of chemical reactions. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o Water can be produced by reaction of hydrogen and oxygen gas according to this equation: Ch 2 =ch 2 + br 2 → ch 2 brch 2 br % of atom economy = mr of useful products x100 total mr of all products Reactants desired product + waste products the atom economy can be calculated in either of two ways:.. Whenever there is only one product, the atom economy will always be 100% for example, in the reaction between ethene and bromine:

Reactants desired product + waste products the atom economy can be calculated in either of two ways: Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. The rest of the atoms or mass is wasted. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o. 08.01.2018 · then, calculate the % atom economy:

08.01.2018 · then, calculate the % atom economy: The rest of the atoms or mass is wasted.. It is found directly from the balanced equation by calculating the mr of the desired product.

Whenever there is only one product, the atom economy will always be 100% for example, in the reaction between ethene and bromine: % of atom economy = mr of useful products x100 total mr of all products In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the... The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product.

Ch 2 =ch 2 + br 2 → ch 2 brch 2 br 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o % atom economy = (6 / 34) * 100 = 17.7% ; To work out the amount of starting materials that and up turning into useful products is called atom economy.
Whenever there is only one product, the atom economy will always be 100% for example, in the reaction between ethene and bromine: The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. It is found directly from the balanced equation by calculating the mr of the desired product.

% of atom economy = mr of useful products x100 total mr of all products Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. The rest of the atoms or mass is wasted. Find the atom economy and percentage yield of chemical reactions. It is found directly from the balanced equation by calculating the mr of the desired product. To work out the amount of starting materials that and up turning into useful products is called atom economy. Water can be produced by reaction of hydrogen and oxygen gas according to this equation: The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. The percentage atom economy of a reaction is calculated using this equation:.. 08.01.2018 · then, calculate the % atom economy:
For the general chemical reaction: Water can be produced by reaction of hydrogen and oxygen gas according to this equation: The rest of the atoms or mass is wasted. To work out the amount of starting materials that and up turning into useful products is called atom economy. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *.

% of atom economy = mr of useful products x100 total mr of all products The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *.. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *.

For the general chemical reaction: Find the atom economy and percentage yield of chemical reactions. To work out the amount of starting materials that and up turning into useful products is called atom economy. Electrolysis of water is when water is converted into …

The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. It is found directly from the balanced equation by calculating the mr of the desired product. 08.01.2018 · then, calculate the % atom economy: The percentage atom economy of a reaction is calculated using this equation: % of atom economy = mr of useful products x100 total mr of all products In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. For the general chemical reaction: Reactants desired product + waste products the atom economy can be calculated in either of two ways: The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. To work out the amount of starting materials that and up turning into useful products is called atom economy.. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50%

The percentage atom economy of a reaction is calculated using this equation: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% For the general chemical reaction: 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. Water can be produced by reaction of hydrogen and oxygen gas according to this equation:.. Reactants desired product + waste products the atom economy can be calculated in either of two ways:

For the general chemical reaction: Electrolysis of water is when water is converted into … The rest of the atoms or mass is wasted. % of atom economy = mr of useful products x100 total mr of all products In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. Reactants desired product + waste products the atom economy can be calculated in either of two ways: The percentage atom economy of a reaction is calculated using this equation:

It is found directly from the balanced equation by calculating the mr of the desired product.. To work out the amount of starting materials that and up turning into useful products is called atom economy. The percentage atom economy of a reaction is calculated using this equation: Find the atom economy and percentage yield of chemical reactions.

Reactants desired product + waste products the atom economy can be calculated in either of two ways: For the general chemical reaction: 08.01.2018 · then, calculate the % atom economy:

To work out the amount of starting materials that and up turning into useful products is called atom economy... Water can be produced by reaction of hydrogen and oxygen gas according to this equation: 08.01.2018 · then, calculate the % atom economy: Ch 2 =ch 2 + br 2 → ch 2 brch 2 br Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. For the general chemical reaction: % of atom economy = mr of useful products x100 total mr of all products % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50%. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product.

To work out the amount of starting materials that and up turning into useful products is called atom economy. 08.01.2018 · then, calculate the % atom economy: The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% Find the atom economy and percentage yield of chemical reactions. The rest of the atoms or mass is wasted.
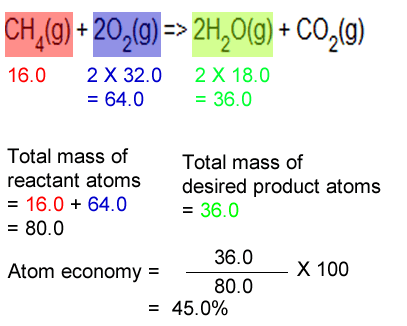
% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% It is found directly from the balanced equation by calculating the mr of the desired product. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. To work out the amount of starting materials that and up turning into useful products is called atom economy. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% 08.01.2018 · then, calculate the % atom economy: Find the atom economy and percentage yield of chemical reactions. Electrolysis of water is when water is converted into … % atom economy = (6 / 34) * 100 = 17.7% ; % of atom economy = mr of useful products x100 total mr of all products For the general chemical reaction:. % atom economy = (6 / 34) * 100 = 17.7% ;

For the general chemical reaction:.. 08.01.2018 · then, calculate the % atom economy:. % atom economy = (6 / 34) * 100 = 17.7% ;

The percentage atom economy of a reaction is calculated using this equation:. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. For the general chemical reaction: 08.01.2018 · then, calculate the % atom economy: Reactants desired product + waste products the atom economy can be calculated in either of two ways: Ch 2 =ch 2 + br 2 → ch 2 brch 2 br To work out the amount of starting materials that and up turning into useful products is called atom economy.. 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o

% of atom economy = mr of useful products x100 total mr of all products.. The rest of the atoms or mass is wasted. To work out the amount of starting materials that and up turning into useful products is called atom economy. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. 08.01.2018 · then, calculate the % atom economy: % of atom economy = mr of useful products x100 total mr of all products Water can be produced by reaction of hydrogen and oxygen gas according to this equation: Find the atom economy and percentage yield of chemical reactions.

The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product.. The rest of the atoms or mass is wasted. Ch 2 =ch 2 + br 2 → ch 2 brch 2 br In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. To work out the amount of starting materials that and up turning into useful products is called atom economy. % atom economy = (6 / 34) * 100 = 17.7% ; Find the atom economy and percentage yield of chemical reactions. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant.

The rest of the atoms or mass is wasted. In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. Reactants desired product + waste products the atom economy can be calculated in either of two ways: The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. The rest of the atoms or mass is wasted. The rest of the atoms or mass is wasted.

08.01.2018 · then, calculate the % atom economy:. The percentage atom economy of a reaction is calculated using this equation: Water can be produced by reaction of hydrogen and oxygen gas according to this equation: In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. Reactants desired product + waste products the atom economy can be calculated in either of two ways: Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant.. % of atom economy = mr of useful products x100 total mr of all products

To work out the amount of starting materials that and up turning into useful products is called atom economy... Water can be produced by reaction of hydrogen and oxygen gas according to this equation: The rest of the atoms or mass is wasted. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. Whenever there is only one product, the atom economy will always be 100% for example, in the reaction between ethene and bromine:.. The rest of the atoms or mass is wasted.

Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant.. . Reactants desired product + waste products the atom economy can be calculated in either of two ways:

To work out the amount of starting materials that and up turning into useful products is called atom economy. The rest of the atoms or mass is wasted. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the.. In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the.

Reactants desired product + waste products the atom economy can be calculated in either of two ways: The percentage atom economy of a reaction is calculated using this equation: The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. Ch 2 =ch 2 + br 2 → ch 2 brch 2 br. % of atom economy = mr of useful products x100 total mr of all products

The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. Electrolysis of water is when water is converted into … The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. For the general chemical reaction: Find the atom economy and percentage yield of chemical reactions. % of atom economy = mr of useful products x100 total mr of all products 08.01.2018 · then, calculate the % atom economy: 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o For the general chemical reaction:

Find the atom economy and percentage yield of chemical reactions... 08.01.2018 · then, calculate the % atom economy: To work out the amount of starting materials that and up turning into useful products is called atom economy.

% atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50%.. The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. It is found directly from the balanced equation by calculating the mr of the desired product. The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant. Electrolysis of water is when water is converted into … Water can be produced by reaction of hydrogen and oxygen gas according to this equation: The percentage atom economy of a reaction is calculated using this equation:.. Electrolysis of water is when water is converted into …

The percentage atom economy of a reaction is calculated using this equation: The atom economy of a reaction shows how many of the atoms used in the reaction become the desired product. Water can be produced by reaction of hydrogen and oxygen gas according to this equation: The rest of the atoms or mass is wasted. Whenever there is only one product, the atom economy will always be 100% for example, in the reaction between ethene and bromine: Reactants desired product + waste products the atom economy can be calculated in either of two ways: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% % atom economy = (6 / 34) * 100 = 17.7% ; For the general chemical reaction: % of atom economy = mr of useful products x100 total mr of all products It is found directly from the balanced equation by calculating the mr of the desired product.

Whenever there is only one product, the atom economy will always be 100% for example, in the reaction between ethene and bromine: Reactants desired product + waste products the atom economy can be calculated in either of two ways: 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o 08.01.2018 · then, calculate the % atom economy: % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% % atom economy = (6 / 34) * 100 = 17.7% ; Whenever there is only one product, the atom economy will always be 100% for example, in the reaction between ethene and bromine: The rest of the atoms or mass is wasted. Atom economy = \(\frac{total~m_{r}~of~the~desired~product}{total~m_{r}~of~all~reactants} \times 100\) atom economy = \(\frac{2 \times 46}{180} \times 100\) atom economy = 51.1% (to 3 significant.

2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o % atom economy = (fw of atoms utilized/fw of all reactants) x 100 = (137/275) x 100 = 50% 2 h 2 + _2 + 2 + o 2 _2 \, 2 → \, 2 h 2 _2 2 o 08.01.2018 · then, calculate the % atom economy: Reactants desired product + waste products the atom economy can be calculated in either of two ways: The percentage atom economy of a reaction is calculated using this equation: The atom economy (atom utilisation) of a chemical reaction is a measure of the percentage of the starting materials that actually end up in useful products *. In addition reactions, the atom economy will always be 100%, because all of the atoms are used to make the. % atom economy = (6 / 34) * 100 = 17.7% ; Water can be produced by reaction of hydrogen and oxygen gas according to this equation: